Historically, claim fees have not been payable at any time during the pendency of a Canadian patent application or term of the patent. This changed on October 3, 2022, and the present article discusses strategies for minimizing claim fees and the potential risks involved.
Summary
As discussed herein:
- Excess claim fees for each claim over 20 are now payable in applications in which examination has been requested on or after October 3, 2022.
- Fees are calculated based on the greatest number of claims pending at any time from when the examination is requested to when the issue fee is paid.
- Claim fees can be avoided or reduced by cancelling claims, making claims multiply-dependent, or by claiming alternatives within a single claim.
- The Supreme Court of Canada has held that a multiply-dependent claim depends on a valid claim and an invalid claim may be valid in so far as it refers to the valid claim.
- A claim that recites alternatives may be invalid if one or more of the alternatives recited in the claim is unpatentable.
- For the avoidance of doubt, embodiments of importance are advantageously claimed separately, even if this incurs claim fees.
Background
Amendments to the
Canadian
Patent Rules introducing excess claim fees came into force recently and apply to any application in which examination was not requested before October 3, 2022. The rules introduce a fee of CAD $100 for each claim
over 20.1 Claim fees are first calculated and payable when the examination is requested. They are calculated again when the final fee (i.e. issue fee) is paid. The excess
claim fee calculation is based on the greatest number of claims pending in the application at any time starting from the date examination is requested and ending on the date the final fee is paid.
This article discusses options and best practices for minimizing claim fees under the new rules. There are three possible approaches for minimizing the number of claims and associated excess claim fees:
- Cancelling claims
- Making use of multiply-dependent claims
- Consolidating subject-matter of separate claims as alternatives within a single claim
Cancelling claims
Given that the USPTO and the EPO permit a maximum of 20 and 15 claims respectively before excess claim fees are payable, applicants may be amenable to prosecuting no more than 20 claims in their Canadian patent applications. Provided the claim set is
reduced to 20 claims prior to or together with the examination request, no claim fees will be payable.
It is important to note that the total claims fees payable will be based on the greatest number of claims pending at any time from when the examination is requested until the final fee (i.e. issue fee) is paid, although the fees are only payable with the examination fee or final fee. Therefore, care must be taken not to unintentionally file a larger claim set at some time during prosecution (e.g. by way of a voluntary amendment or in response to an Examiner’s Report) even if such claims are not ultimately examined or granted. The following example illustrates the consequences of filing excess claims during prosecution:
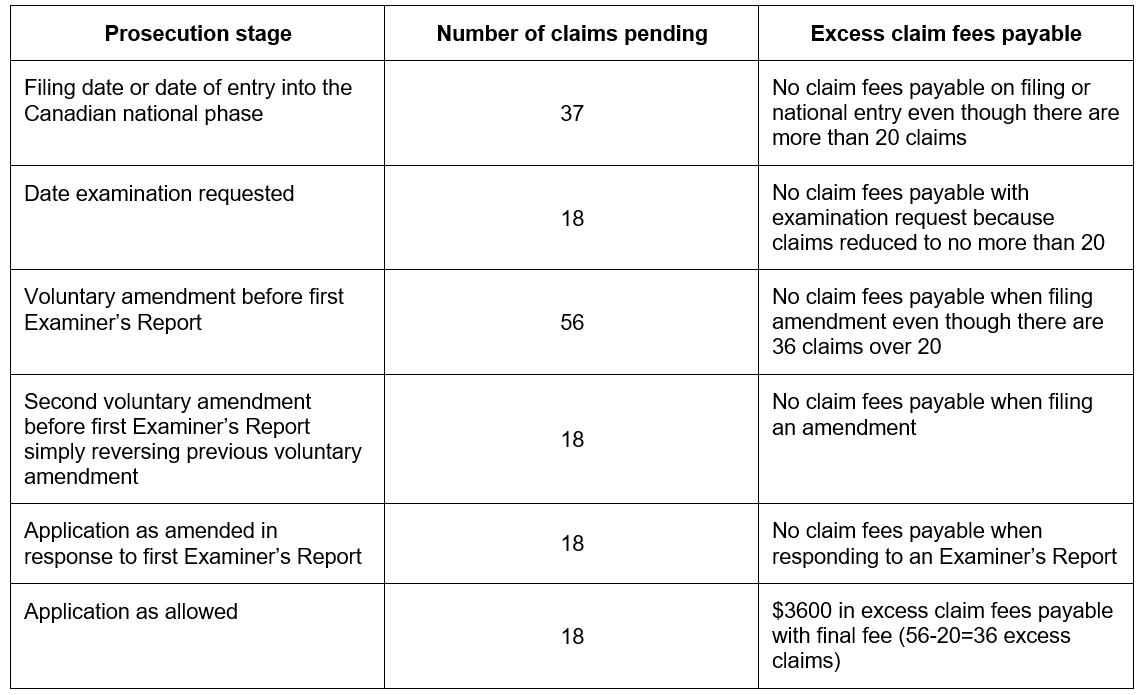
In the above example, although only the 18 claims originally presented for examination were ultimately examined, allowed, and granted, excess claim fees of CAD $3600 were payable when the final fee was paid in view of the set of 56 claims that were pending
briefly after the examination was requested and before substantive examination commenced.
Illustrative claim set
The following illustrative claim set will be used throughout the remainder of this article to illustrate claim fee calculations and matters of claim interpretation and validity:
- A pharmaceutical composition comprising compound A and a pharmaceutically acceptable excipient.
- A pharmaceutical composition comprising compound B and a pharmaceutically acceptable excipient.
- A pharmaceutical composition comprising compound C and a pharmaceutically acceptable excipient.
- The pharmaceutical composition according to any one of claims 1 to 3, in the form of a tablet.
- The pharmaceutical composition according to any one of claims 1 to 3, in the form of a capsule.
- The pharmaceutical composition according to any one of claims 1 to 5, wherein said pharmaceutically acceptable excipient comprises:
- a flavouring agent
- a disintegrant,
- a colourant,
- a glidant, or
- a preservative.
- The pharmaceutical composition according to any one of claims 1 to 6, comprising, as a non-nutritive sweetener, a sugar alcohol selected from the group consisting of:
- xylitol,
- maltitol,
- sorbitol,
- mannitol, and
- erythritol.
- The pharmaceutical composition according to any one of claims 1 to 7, comprising, as a preservative, an antioxidant that is:
- butylated hydroxyanisole (BHA),
- butylated hydroxytoluene (BHT),
- sodium metabisulfite (SMB), or
- propyl gallate (PG).
Multiply-dependent claims
Multiply-dependent claims (claims 4-8 in the illustrative claim set) are permitted, and a multiply-dependent claim may depend on another multiply-dependent claim (claims 6-8 in the illustrative claim set). In contrast to US practice, in Canada, a multiply-dependent
claim counts only as a single claim for the purposes of calculating claim fees, the relevant provisions of the rules providing:
A claim that defines the subject-matter of an invention in the alternative, including a dependent claim within the meaning of section 63 that refers to more than one preceding claim, counts as a single claim for the purposes of [claim fee calculations].2
The illustrative claim set, therefore, contains only 8 claims for fee calculation purposes. Claim fees would accrue quickly if the multiply-dependent claims were rewritten with only single dependencies. For instance, each of claims 4 and 5 could be written
as 3 singly-dependent claims, claim 6 written as 9 singly-dependent claims, claim 7 written as 18 singly-dependent claims, and claim 8 written as 36 singly-dependent claims. Thus, to obtain the same coverage without the use of multiple claim dependencies,
the illustrative claim set would have to contain 72 rather than 8 claims for fee calculation purposes.
Pursuant to section 58 of the Patent Act, invalid claims in a patent do not affect valid claims:
When, in any action or proceeding respecting a patent that contains two or more claims, one or more of those claims is or are held to be valid but another or others is or are held to be invalid or void, effect shall be given to the patent as if it contained
only the valid claim or claims.
In contrast to the new fee calculation provisions, the Patent Rules describe each claim dependency within a multiply-dependent claim as a separate claim.
Subsection 63(3) of the Patent Rules provides that:
A dependent claim that refers to more than one claim must refer to those claims in the alternative only.
Subsection 63(4) of the Rules provides that:
A dependent claim is considered to include all the limitations contained in the claim to which it refers or, if the dependent claim refers to more than one claim, any particular alternative of the dependent claim is considered to include all the limitations
contained in the particular claim in respect of which it is considered.
Moreover, in Consolboard,3 without referencing any particular provision of the Patent Act or
Patent Rules,4 the Supreme Court of Canada held that dependent claim 10 of Canadian Patent No. 565,618 directed to “A consolidated structure as claimed in claim 7, 8 or 9, in which the tapered ends of the wafers are ragged,”
was valid “
in so far as claim 10 includes claim 7.” Each of claims 7, 8, and 9 were independent claims to a “consolidated structure,” namely a wafer board composed of woody wafers and a thermosetting resinous binder. Independent claim
7 was found to be valid but claims 8 and 9 invalid. Multiply-dependent claim 10 was valid when dependent on claim 7. The Consolboard approach, as it applies to multiply-dependent claims, has been followed by the Federal Court in G.D. Searle.5 Multiply-dependent claims have thus been found to constitute separate claims for the assessment of validity.
There are other dependent claim formats that might be employed to reduce claim fees and that have not been specifically considered by the courts or that are simply uncommon.
A claim in the form “16. A compound according to any one of claims 1-5, a pharmaceutical composition according to any one of claims 6-10, or a kit according to any one of claims 11-15, wherein R1 is -OH” has not routinely
been used in Canada but could in principle be employed to minimize claim fees. Even though the preamble of the claim refers to preceding claims in different categories, reference is made to each claim separately and the claim meets all the formal
requirements for a dependent claim set out in section 63 of the Patent Rules. Consistent with the finding of the Supreme Court in Consolboard, it appears claim 16 should be considered separately with respect to each of its dependencies.
For instance, if claim 5 to a compound were invalid but claim 12 to a kit valid, claim 16 could be valid in so far as it depends from, and further limits, claim 12 directed to a kit.
A commonly used claim format is “17. Use of a compound according to any one of claims 1-5 for the treatment of diabetes.”
This claim differs from previous examples in that the claim is directed to use and refers to preceding claims directed to compounds. The claims are thus in different “categories,” in contrast to the claims in the illustrative claim set above.
Referencing somewhat different examples, the Manual of Patent Office Practice (MOPOP) states that claims of one category referring to claims of another category may be permitted, but are not dependent claims:6
Claims are also permitted to refer to other claims or parts of claims of the same or of another category, in order to avoid repeating lengthy definitions already given and to simplify claiming, provided they do not become ambiguous as a result of such
dependency, thereby contravening section 27(4) of the Patent Act. Such claims however are not dependent claims and section 63 of the Patent Rules does not apply. The patentability of the claim referred to does not
necessarily imply the patentability of the dependent claim containing the reference.
[emphasis added]
However, the MOPOP is not a binding legal authority, and no authority is cited for this proposition. Indeed, exemplary claim 17 appears to meet all the requirements of a dependent claim set forth in subsections 63(1)-(3) of the Patent Rules:
- It “includes all the features of one or more other claims” in that it does not cover the use of any compounds not encompassed by the compound claims to which it refers.
- It refers “by number” to the other claim or claims.
- It states “the additional features claimed.” In this regard, the additional feature is the limitation to the use of the compound for treating diabetes. The preceding claims are infringed by the use of the compound for any purpose, whereas claim 17 is limited in scope to use in the treatment of diabetes.
- It refers only to “a preceding claim or claims.”
- It refers to claims 1-5 “in the alternative only.”
As it seems to meet the prescribed formal requirements, it appears that exemplary claim 17 reasonably should be construed as a dependent claim pursuant to subsection 63(4) of the Patent Rules and therefore be:
…considered to include all the limitations contained in the claim to which it refers or, if the dependent claim refers to more than one claim, any particular alternative of the dependent claim is considered to include all the limitations contained
in the particular claim in respect of which it is considered.
Nevertheless, for the avoidance of any doubt, the most cautious course is to include a separate claim for subject-matter of particular importance, even if excess claim fees may be incurred.
Claims reciting “alternatives”
Claims 6-8 in the illustrative claim set each recites a group of elements. For the purposes of the claim fee rules, it is expected that the elements within the claims should constitute “alternatives” that do not count towards claim fees,
such that each of claims 6-8 indeed constitutes a single claim. As discussed above, the relevant rules provide:
A claim that defines the subject-matter of an invention in the alternative, including a dependent claim within the meaning of section 63 that refers to more than one preceding claim, counts as a single claim for the purposes of [claim fee calculations].7
The question then arises as to the validity of the claim if one more (but not all) of the alternatives recited in the claim is found to be unpatentable. Is the claim valid to the extent it recites only the patentable alternative(s)?
Subsection 27(5) of the Patent Act, which has been cited in connection with this issue, provides:
For greater certainty, if a claim defines the subject-matter of an invention in the alternative, each alternative is a separate claim for the purposes of sections 2, 28.1 to 28.3, 56 and 78.3.
The provision is difficult to understand, particularly in view of the referenced sections of the Patent Act. Section 2 is the definition section, including the definition of “invention” as “any new and useful, art, process,
machine, manufacture, or composition of matter [or improvement therein].” Section 28.1 concerns the “claim date” (i.e. priority date for each claim). Section 28.2 is the novelty provision. Section 28.3 is the obviousness provision.
Section 56 concerns prior user rights. Finally, section 78.3 is a transitional provision concerning the application of “first to invent” rules under the Patent Act as it read prior to October 1, 1989. This is indeed a perplexing
collection of seemingly unrelated provisions.
In the context of reciting alternatives within a claim to reduce claim fees, subsection 27(5) raises two questions:
- When does a claim define subject-matter “in the alternative” such that subsection 27(5) applies?
- If subsection 27(5) applies, what are the consequences?
Only a few court decisions have interpreted subsection 27(5) and the issues are commingled. It is helpful briefly to discuss each of the cases.
As a preliminary matter, the illustrative claim set above provides examples of claims that may or may not define subject-matter “in the alternative”. The various pharmaceutically acceptable excipients in claim 6 cannot be substituted for each
other interchangeably — a glidant cannot be used in place of a flavouring agent to achieve the same result. And the members of the list are separated with the disjunctive term “or.” In contrast, claim 7 recites a list of sugar alcohols
that are non-nutritive sweeteners. It can be assumed that one could be used in place of another in a pharmaceutical composition to achieve the same result. Moreover, the claim uses the ostensibly conjunctive expression “selected from the
group consisting of A, B, C…and D.” Finally, claim 8 is substantively similar to claim 7 (the various recited antioxidants could be substituted for each other as preservatives) but the elements of the list are again separated
by “or.”
In practice, the Canadian Patent Office accepts claims in any of these formats, and provides specific guidance with respect to so-called “Markush claims” (i.e. the format of claim 7) in the MOPOP:8
In chemical cases, a claim directed to a genus expressed as a group consisting of certain specified materials is allowable (Ex parte Markush 1925, 340 U.S.O.G. 839) provided it is clear from the known nature of the alternative materials or from the prior art that the materials in the group possess at least one property in common which is mainly responsible for their function in the claimed relationship. Therefore, a Markush claim will generally be construed with a generic expression covering a group of two or more different materials (elements, radicals, compounds) as illustrated in the following examples:
A solvent selected from the group consisting of alcohol, ether and acetone...
A strip of a conductive metal selected from the group consisting of copper, silver and aluminium...
Occasionally, the Markush format may be used in claims directed to the subject matter in the mechanical or electrical fields in a manner such as that illustrated in the example below:
A means for attaching a wall panel to a framework wherein the attaching means is selected from a group consisting of nails, rivets and screws...
[emphasis added]
Claim 6 in the illustrative claim set does not meet the criteria of a “Markush claim” as described above because the recited elements do not possess a property in common and they do not serve the same function in the claimed invention. In
contrast, despite the grammatical differences (“or” versus “selected from the group consisting of…and”), it would seem that, substantively, both claims 7 and 8 should meet the requirements for a Markush claim because
the elements in the lists constitute functionally equivalent alternatives that may be substituted for each other in the context of the claimed invention.
In Abbott I,9 a claim to a method for making Clarithromycin Form II involving use of a solvent “selected from the group consisting of acetone,…, and methanol-ethanol”
was held to be a Markush claim, such that each recited solvent must be capable of being used to produce Clarithromycin Form II. The Federal Court concluded that the solvents were not “alternatives” within the meaning of subsection 27(5),
and therefore subsection 27(5) did not apply. Because the utility of only some but not all the solvents had been established, the Court considered that the entire claim failed.
In Abbott II,10 another proceeding concerning the same patent, the Federal Court agreed that the relevant claim was a Markush claim in which each recited
solvent can be used to produce Clarithromycin Form II, and the claim did not recite “alternatives” for the purposes of subsection 27(5). The Court further stated that subsection 27(5) is not a saving or remedial
provision but rather an “administrative provision for purposes of a patent application” such that if any one alternative in a claim is not patentable, the entire claim fails, finding it particularly significant that subsection 27(5) did
not reference section 58 of the Patent Act (the provision under which invalid claims can be severed from valid claims). The Court moreover noted that a claim could recite a vast number of alternatives, and the adverse consequences of overclaiming
exclude the competing interpretation that subsection 27(5) would save a claim if at least one of the alternatives is patentable. Thus, even had the claim at issue recited “alternatives” within the meaning of subsection 27(5), the Court
was of the view that this provision would not have saved the claim because not all alternatives were patentable.
G.D. Searle11 considered subsection 27(5) but in the context of a multiply-dependent claim. As discussed above, consistent with the finding of the Supreme
Court in
Consolboard, the Federal Court held that the language “any of claims 1-7” in multiply-dependent claim 8 in the patent in question could be construed as claim 8 referring only to claim 4, and the validity of claim 8 considered
only in so far as it referred to claim 4, not claims 1-3 or 5-7.
Schering-Plough12 considered a claim reciting a composition being in “one of tablet or capsule form.” The Federal Court held that the entire
claim failed because the capsule (but not the tablet) was anticipated by the prior art. Without discussing whether tablets and capsules were functionally equivalent or interchangeable, the Court held that they were “alternatives,” but
that the alternative claim to a tablet was not saved by subsection 27(5), following the interpretation of 27(5) in Abbott II.
In Eli-Lilly,13 the Federal Court took a different approach in applying 27(5) to a claim directed to the use of a compound “selected from the group consisting of [tadalafil] and [3-methyl tadalafil]”
for the treatment of erectile dysfunction. The Court held that, if a claim recites alternatives, under subsection 27(5) the entire claim fails if one of the alternatives is invalid. This is consistent with Abbott II and
Schering-Plough, holding that 27(5) is not a remedial provision. The Court concluded that 27(5) did not apply because the claim did not recite “alternatives,” but was instead a Markush claim, in which the two recited compounds
both could be used to achieve the desired result of treating erectile dysfunction. As the utility of at least tadalafil had been established for this purpose, it was irrelevant and of no consequence whether the utility of 3-methyl tadalafil had also
been established.
On appeal14 the Federal Court of Appeal found that this reasoning was in error. Citing Abbott I, but without referencing 27(5), the Court held that a Markush claim
requires that “each compound in the claimed class, not merely one of the compounds, have utility.” The Court noted that the matter was of no consequence on the facts, finding that the utility of both tadalafil and 3-methyl tadalafil
had been established in any event.
Based on the case law to date, the cautious view is that each of claims 6-8 in the illustrative claim set may fail in its entirety if one of the claimed elements is found unpatentable on the basis that:
- A “Markush” claim that recites a group of elements that have a common property and can be used to achieve the same result does not recite “alternatives” within the meaning of subsection 27(5) of the Patent Act.
- Each recited member of the group must be patentable for a Markush claim to be valid.
- If the claim does not recite a Markush group and the elements are true “alternatives” within the meaning of subsection 27(5) such that the provision applies, then subsection 27(5) is not a remedial provision and its effect is that the claim fails if any one alternative in the claims is unpatentable.
Conclusion
Excess claim fees may be reduced or avoided by using multiple claim dependencies and by reciting alternatives within a claim. Multiple claim dependencies do not appear to raise validity concerns as the Patent Rules expressly provide that a dependent claim is considered to include all the limitations contained in the particular claim in respect of which it is considered, and the Supreme Court has held that a multiply-dependent claim depending from both valid and invalid claims is valid in so far as it is considered with respect to the valid claim from which it depends. In contrast, reciting alternatives within a claim, whether in the form of a Markush group or as true alternatives, may result in the claim being found invalid if any one of the alternatives is unpatentable.
Ideally, a patent application would include a species or picture claim to any embodiment of commercial importance, even if excess claim fees are incurred. The government fee of CAD $100 for such a claim is a small price to pay if it avoids any argument that the claim is potentially invalid as being overbroad.
If you have questions or require further information, please contact a member of the Patents group at Smart & Biggar.
The preceding is intended as a timely update on Canadian intellectual property and technology law. The content is informational only and does not constitute legal or professional advice. To obtain such advice, please communicate with our offices directly.
References
1. No distinction is drawn between independent and dependent claims for the purposes of excess claim fees. The fee is CAD $100 for each claim over 20.
2. This language is found in both subsection 80(1.1) (excess claim fees when requesting examination) and subsection 87(1.1) (excess claim fees payable with final fee) of the Patent Rules.
3. Consolboard Inc v MacMillan Bloedel (Sask.) Ltd, [1981] 1 SCR 504 at pp. 537-538.
4. Including section 60 of the Patent Act, R.S.C. 1970, chapter P-4, the previous counterpart to current section 58.
5. G.D. Searle & Co v Novopharm Limited, 2007 FC 81 (celecoxib) at para [26]-[32].
6. Manual of Patent Office Practice (MOPOP) at §16.06. Notably, the MOPOP is not a binding legal authority. As stated in §1.01: “This manual is a guide only and should not be considered legally binding. If there are inconsistencies between the information in this manual and the applicable legislation, the legislation must be followed. The information provided is for information purposes only and should not be relied upon for legal purposes or business decisions.”
7. Patent Rules, supra note 2.
8. MOPOP §16.11. As per note 6, the MOPOP is not a binding legal authority.
9. Abbott Laboratories and Abbott Laboratories Limited v The Minister of Health and Ratiopharm, a division of Ratiopharm Inc, 2005 FC 1095 (clarithromycin) at para [20]-[33].
10. Abbott Laboratories and Abbott Laboratories Limited v The Minister of Health and Apotex Inc, 2005 FC 1332 (clarithromycin) at para [37]-[57].
11. G.D Searle, supra, note 5.
12. Schering-Plough Canada Inc and Schering Corporation v Pharmascience Inc, Sepracor Inc and the Minister of Health, 2009 FC 1128 (desloratadine) at para [88]-[93].
13. Eli Lilly Canada Inc v Mylan Pharmaceuticals ULC, The Minister of Health, and ICOS Corporation, 2015 FC 17 (tadalafil) at para [116]-[120].
14. Mylan Pharmaceuticals ULC v Eli Lilly Canada Inc, ICOS Corporation, and The Minister of Health, 2016 FCA 119 (tadalafil) at para [56].
Related Publications & Articles
-
Adeia Guides Inc v Videotron Ltd: infringement through common design – a Canadian first
In the latest matter concerning Adeia Guides Inc’s (“Adeia”, formerly “Rovi Guides Inc”) patent enforcement campaign against Canadian cable service providers, the Federal Court held, for the first tim...Read More -
Not so nominal: damages available for brand owners facing evasive counterfeiters
Companies invest significant resources in building consumer awareness and goodwill in their brands. Over time, strong brands come to represent more than just a product name or logo; they may signal a ...Read More -
Brand protection online: Enforcement options for domain name takedowns
A well‑chosen domain name is a valuable brand asset. It serves as a powerful marketing tool by helping businesses stand out in a crowded digital landscape and attracting consumers to their websites. W...Read More
