The interim approach proposed a “status quo” to pricing until Guidelines could be established relating to the final regulations. As we previously reported, the final regulations included the new “basket” of foreign reference countries but repealed the provisions relating to: (i) additional price regulatory factors and related reporting requirements, and (ii) the collection of price and revenue information net of third-party rebates. Neither the current Guidelines (last updated in February 2017) nor the 2020 Guidelines address the final regulations.
The Board had invited comments on the proposed interim approach to pricing and has published the 37 submissions received.
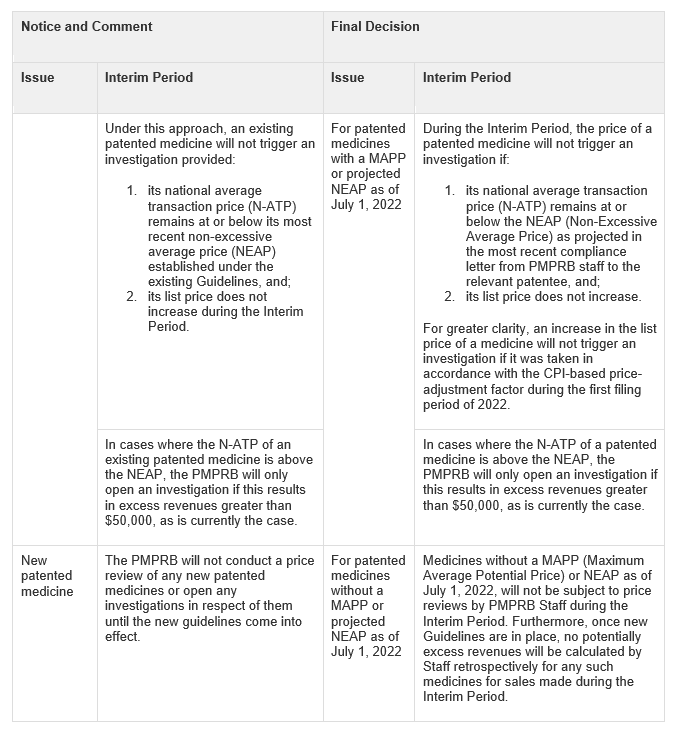
A final decision on the interim approach was announced on August 18, 2022. The Board has largely maintained its proposed approach, noting the following distinctions between the original proposal and the final decision:
The announcement also includes FAQs addressing the questions:
- How can patentees know what the NEAP is that will apply to the price of a medicine during the Interim Period?
- Will the NEAPs continue to apply to the price of medicines if new Guidelines are not in place by the end of 2022?
- How will the PMPRB address the value of free and compassionate goods during the Interim Period?
- Will HDAP meetings continue on their current timelines for new (and future) medicines?
Guideline consultations are expected to begin in September of this year, and the Board has committed to completing the consultation process and issuing new Guidelines before January 1, 2023.
In the meantime, should you have any questions, please do not hesitate to contact a member of the Life Sciences Regulatory & Compliance Group.
The preceding is intended as a timely update on Canadian intellectual property and technology law. The content is informational only and does not constitute legal or professional advice. To obtain such advice, please communicate with our offices directly.
Related Publications & Articles
-
Canada announces new Pharmaceutical and Life Sciences Sector Task Force
On March 18, 2026, the federal Minister of Health announced the creation of the Pharmaceutical and Life Sciences Sector Task Force, a joint initiative led by Health Canada and Innovation, Science and ...Read More -
Canada’s Drug Agency releases updated Procedures for Reimbursement Reviews & new data on Health Technology Assessment timelines
On April 30, 2026, Canada’s Drug Agency (CDA) published updated Procedures for Reimbursement Reviews, following completion of a public consultation earlier this year. The updated procedures will be ef...Read More -
PMPRB releases May NEWSLetter and Meds Pipeline Monitor
On May 12, 2026, the Patented Medicine Prices Review Board (PMPRB) released its May NEWSLetter. The NEWSletter provides updates on the implementation of the new Guidelines for PMPRB Staff, the consult...Read More
