September 21, 2021 marked the fourth anniversary of the significant amendments to the Patented Medicines (Notice of Compliance) Regulations (Regulations). This article provides an update on activities in the fourth year following the amendments, including new actions and a wave of Court decisions, both on the merits and procedural (see our prior first, second and third anniversary updates).
Status of actions under the Regulations
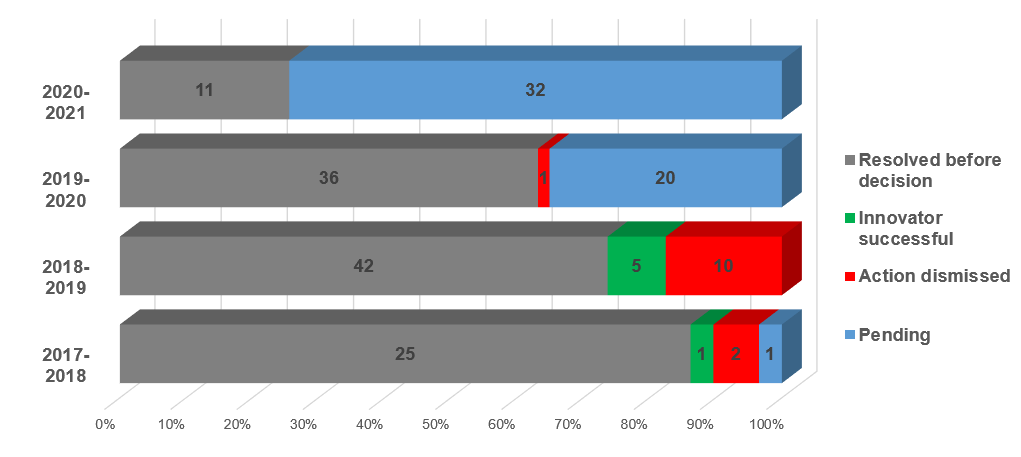
In the period of September 21, 2020, to September 21, 2021, approximately 30 sets1 (38 total) of actions were started.* This is a drop from the last two years (49 in 2019-2020 and 35 in 2018-2019).
Of the approximately 181 actions* started since the amendments, the majority were resolved before a decision on the merits (114, in grey below), 19 actions were pursued to a decision on the merits, with the majority of actions dismissed (13, in red below). Appeals are pending from 18 actions; one trial decision was affirmed on appeal.
(*Corrected September 27, 2022: The original number included five section 8.2 actions.
The graph below has not been revised.)
The following is a list of drugs for which there is at least one active section 6 action (although some of the actions may be stayed or adjourned pending appeal):
- Adalimumab (Abbvie’s HUMIRA)
- Generic: JAMP
- Apixaban (BMS’s ELIQUIS)
- Generic: Sun Pharma
- Canagliflozin (Janssen’s INVOKANA)
- Generic: Sandoz
- Dolutegravir (Viiv’s TIVICAY)
- Generic: Sandoz
- Dapagliflozin (AstraZeneca’s FORXIGA)
- Generics: Sandoz; Teva
- L-lysine-d-amphetamine (Shire’s VYVANSE)
- Generic: Apotex (stayed pending appeal of proceedings under pre-amended Regulations)
- Linagliptin (Boehringer Ingelheim’s TRAJENTA)
- Generics: Teva; Sandoz; JAMP
- Lurasidone hydrochloride (Sunovion’s LATUDA)
- Generics: Pharmascience; Taro; Jamp
- Macitentan (Janssen’s OPSUMIT)
- Generics: Apotex; Jamp; Sandoz
- Naloxone hydrochloride (Emergent’s NARCAN Nasal Spray)
- Generic: Teva
- Paliperidone palmitate (Janssen’s INVEGA SUSTENNA)
- Generics: Pharmascience; Apotex
- Perampanel (Eisai’s FYCOMPA)
- Generic: Taro
- Rivaroxaban (Bayer’s XARELTO)
- Generic: Pharmascience
- Sapropterin dihydrochloride (BioMarin’s KUVAN)
- Generic: Dr. Reddy’s Laboratories
- Saxagliptin (AstraZeneca’s ONGLYZA)
- Generic: Sandoz
- Sitagliptin (Merck’s JANUVIA)
- Generics: Dr. Reddy’s Laboratories; Teva; Accord; Sandoz; Pharmascience; Apotex; JAMP
- Sitagliptin and metformin hydrochloride (Merck’s JANUMET and JANUMET XR)
- Generics: Dr. Reddy’s Laboratories; Teva; Sandoz; Apotex
- Sunitinib malate (Pfizer’s SUTENT)
- Generic: JAMP
- Tofacitinib (Pfizer’s XELJANZ)
- Generic: JAMP
- Vortioxetine (Lundbeck’s TRINTELLIX)
- Generic: Apotex
Over the last four years, Sandoz was the most active patent challenger (defendant in 47 actions relating to 23 drugs), followed by Apotex (29 actions relating to 17 drugs) and Teva (20 actions relating to 15 drugs).
In the past year, there were also actions commenced further to section 8.1, which permits “an interested person” to bring an action for the declaration that the patent/certificate of supplementary protection or any claim is invalid or void, and section 8.2, which permits a first person/patentee to bring an infringement action regarding a patent not listed on the Patent Register once served with a Notice of Allegation (NOA):
- Section 8.1
- 4 actions relating to adalimumab (Abbvie’s HUMIRA) commenced by JAMP
- Section 8.2
- 4 actions relating to Merck’s sitagliptin (JANUVIA) and
- 1 action relating to Merck’s sitagliptin / metformin (JANUMET XR)
Several actions have also been commenced in the past year pursuant to section 8 of both the amended and pre-amended Regulations, seeking damages following unsuccessful patent litigation:
- Abiraterone acetate (commenced by each of Dr. Reddy’s Laboratories and Apotex)
- Silodosin (commenced by Sandoz)
- Pirfenidone (commenced by Sandoz)
- Amlodipine and perindopril (pursuant to pre-amended Regulations, commenced by Apotex)
The first (and only other) amended section 8 action which was commenced by Pfizer in 2020, seeking damages after Amgen was unsuccessful in filgrastim litigation, was discontinued following the pleadings stage. The first amended section 8 trial (relating to Apotex’s abiraterone) is scheduled for June 2023.
Trial Decisions on the Merits
A number of decisions on the merits were released in the fourth year, including as set out in our mid-year highlights:
- Allergan v Sandoz (silodosin, RAPAFLO) –claims to be a capsule of silodosin that achieves a defined rapid dissolution profile valid but not infringed.
- Teva v. Pharmascience (glatiramer acetate, COPAXONE, GLATECT) – Claims to use of glatiramer acetate in treating patients who have had a single clinical attack suggestive of multiple sclerosis before confirmed diagnosis (437 patent) invalid (not anticipated but obvious); claims to use of 40 mg glatiramer acetate administered by subcutaneous injection three times per week for treatment of relapsing-remitting multiple sclerosis (802 patent) valid (not obvious, useful) and infringed.
- Bristol-Myers Squibb v. Pharmascience, Sandoz (apixaban, ELIQUIS) – Claims to apixaban and use of apixaban in treatment of thromboembolic disorders (202 patent) valid (attacks included inutility of selection) and infringed; claims to the formulation of 2.5 and 5 mg apixaban (dry granulation) tablets (171 patent) valid (attacks included obviousness) and infringed.
- Janssen v. Pharmascience (abiraterone acetate, ZYTIGA) – Claims to use of a combination of abiraterone acetate and prednisone for the treatment of prostate cancer invalid (obvious).
- Hoffmann-La Roche v. Sandoz (pirfenidone, ESBRIET) – Pirfenidone for use in a specific dose escalation regimen (654 patent) and for use at a specific dose in a patient that experiences a specific liver abnormality (997 patent) invalid (method of medical treatment and obvious).
Appeal Decisions on the Merits
The Federal Court of Appeal released two decisions upholding the lower Court’s decisions granting applications for Orders of prohibition:
- Apotex v Janssen (abiraterone acetate, ZYTIGA) – Prohibition Order under pre-amended Regulations was affirmed by the FCA. Claims to use of abiraterone acetate and prednisone for treatment of prostate cancer valid (patentable subject matter (synergy not required), not obvious, useful) and infringed (Note opposite result following a trial under the amended Regulations – see trial decisions on the merits above).
- Apotex v. Shire (lisdexamfetamine, VYVANSE) – Prohibition Order under pre-amended PMNOC Regulations / dismissal of impeachment action affirmed by FCA. Claims to lisdexamfetamine valid (not anticipated based on disclosure of genus, not obvious). Apotex has sought leave to appeal to the Supreme Court.
Additionally, the Federal Court of Appeal dismissed Amgen’s appeal of the trial judge’s finding that Amgen’s patent claiming G-CSF made using recombinant genetic technology was obvious. Amgen’s leave to appeal to the Supreme Court was also dismissed.
Procedural Matters
As set out in our mid-year highlights:
- A defendant was permitted to amend its statement of defence to plead validity attacks beyond its notice of allegation; decision was upheld on appeal.
- Section 8.2, which permits a claim on an unaddressed patent, e.g. an unlisted patent, once a NOA is served, does not impose a deadline for commencing an action.
- A re-examination proceeding was stayed pending Pharmascience’s appeal of a PMNOC judgment finding the patent valid and infringed.
- A plaintiff was denied leave to amend its statement of claim to plead infringement of further patents more than 45 days after receipt of NOAs.
Conclusion
The fourth year brought further decisions on the procedure and an influx of new decisions on the merits. We will continue to report on further developments in the coming year.
Should you have any questions, please do not hesitate to contact a member of the Pharmaceutical Litigation group.
References
1. Actions between the same parties in respect of the same innovator reference drug are considered one “set”. The statistics are provided as of the fourth-year anniversary, September 21, 2021.
Related Publications & Articles
-
PMPRB update: Guidelines for PMPRB Staff come into effect January 1, 2026; 2024 Annual Report
On January 1, 2026, the Guidelines for PMPRB Staff will come into effect. In November, the Patented Medicine Prices Review Board (PMPRB) held information sessions on the Guidelines to share technical ...Read More -
2025 highlights in Canadian life sciences IP and regulatory law
In 2025, the Rx IP Update team at Smart & Biggar reported on a number of developments in Canadian life sciences IP and regulatory law.Read More -
